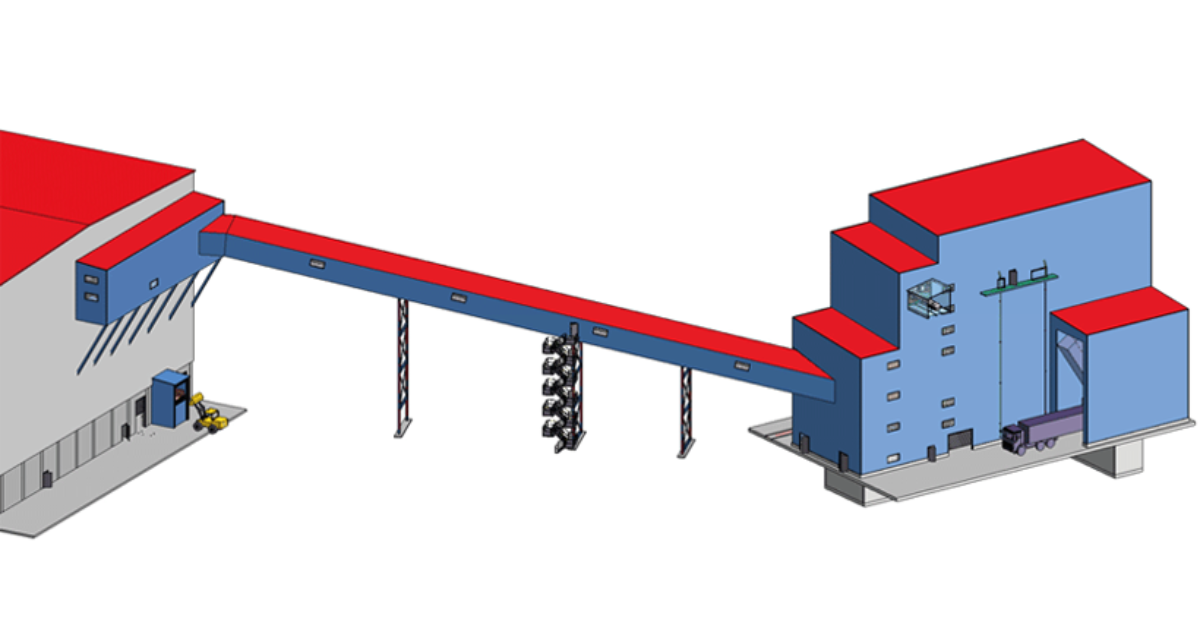
Arglass and Zippe Join Forces for a More Efficient and Sustainable Glass Production in Valdosta, USA
Arglass and Zippe are joining forces to construct a second Arglass plant in Valdosta, USA. The project is set to be completed in December 2024 and will be commissioned in…
JD Sports Looks to Expand in the US with Acquisition of Hibbett Inc Amid Tough Competition in Global Sportswear Market
JD Sports Fashion, a UK-based sportswear retailer, has proposed to buy US athletic fashion retailer Hibbett Inc for around $1.08 billion. The move is strategic as JD Sports aims to…
Spacecraft Spitzer’s Autonomous Safety Systems Updated for Unprecedented Orbit Challenges
As Spitzer’s orbit progresses beyond its current phase, it presents new challenges for the spacecraft. The distance between the two planets is increasing due to the slower pace of Spitzer’s…
RiverStone Health and Gainan’s Heights Flowers Partner to Offer Free Soil Lead Testing in Yellowstone County
As a journalist, I have rewritten the given article in a unique way. In Yellowstone County, despite the presence of lead in all parts of the environment, including air, water,…
Economic Prosperity and Political Concerns: The Impact of EU Enlargement on Central and Eastern Europe
Central and Eastern Europe has experienced remarkable economic growth since joining the European Union, with countries such as Poland, Hungary, Slovakia, Slovenia and Czech Republic accounting for 8.7% of EU…
Jerry Seinfeld on the State of the Movie Industry: A Chat with GK
In an interview with GK, Jerry Seinfeld recently shared his insights on the current state of the movie business. According to the comedian, there is a sense of confusion among…
Israeli Army Intelligence Chief Resigns After Hamas Attack: A Precedent Set for Accountability in National Security Matters?
In the aftermath of the events of October 7, Israeli army intelligence chief Aharon Haliva announced his resignation. He took responsibility for the attacks carried out by Hamas, which resulted…
CTO Aaron LaBerge Steps Down from Disney and Picks Up New Role in Gaming: A Look at his Legacy and Future
Aaron LaBerge, who has served as chief technology officer at Disney Entertainment and ESPN for over 20 years, is stepping down from the company. In an internal memo to staff,…
Chris Paul’s Future Uncertain: The Warriors May Lose Out on Signed Point Guard for Financial Reasons
Golden State Warriors fans may be disappointed to hear that point guard Chris Paul is unlikely to return to the team next season due to financial reasons. While Paul’s contributions…
Franklin Science Center Evacuated Due to Suspected Burning Incident: Authorities Investigate Cause of Odor
On Monday afternoon, the Franklin Science Center was evacuated due to the smell of something burning detected in the building. At 3:24 p.m., SU News sent out a mass email…